This goal is intended to improve the historical lack of efficiency of clinical trials and the current development paradigm for new drugs that has dramatically increased research and development spending while producing fewer new drugs (Woodcock and Woosley 2008). A stated goal of the FDA has been that all future clinical trials will be designed using model-based simulations to improve the success rate in clinical trials and to improve the efficiency and probability of obtaining informative results from clinical trials (Gobburu 2010).
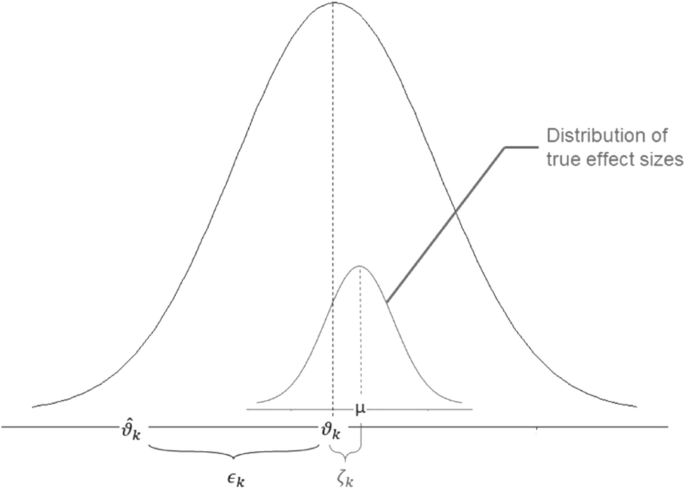
The ability to simulate from pharmacometric models is a very powerful tool for informed decision making. Caution must be used in the extrapolation of models to new circumstances since the assumptions of the model include the nature of the data on which they were built. These predictions may include other experimental design characteristics or other patient characteristics. Extrapolation is used to predict values outside the range of conditions on which the model was built. For instance, exposures at a dose of 150 mg may be interpolated using a model that was built on data obtained only from doses of 100 and 200 mg. Interpolation predicts new values within the range of conditions on which the model was built. Predictive models may be used for interpolation or extrapolation. Predictive model applications generally follow and build upon the development of descriptive models. Predictive models enable model-based simulations for testing conditions for which observed data are not available to the researcher. Descriptive models are used to characterize existing data. Models may be descriptive or predictive in their applications. Hypotheses may be developed and tested with models, which may lead to changes in behaviors, such as altering a dosing regimen based on an observable or measurable feature of a patient, such as body weight. Models can be used to explore mechanisms or outcomes of a system, whether that is a physiological process, drug action, disease progression, or the economic impact of a particular therapy. These models are used to concisely summarize the behavior of large quantities of data using a small number of numerical values and mathematical expressions.

Pharmacometrics is a science that employs mathematical models in diverse applications of the pharmaceutical sciences. Population Model Concepts and Terminology
